Polyethylene or polyethene is a thermoplastic commodity heavily used in consumer products (over 60M tons are produced worldwide every year). Its name originates from the monomer ethene used to create the polymer.
In the polymer industry the name is sometimes shortened to PE, similar to how other polymers like polypropylene and polystyrene are shortened to PP and PS, respectively. In the United Kingdom the polymer is called polythene. (e.g. in the Beatles song Polythene Pam).
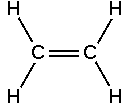
The ethene molecule (known almost universally by its non-IUPAC name ethylene), C2H4 is CH2=CH2, Two CH2 connected by a double bond, thus:
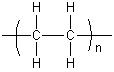
Polyethylene is created through polymerization of ethene. It can be produced through radical polymerization, anionic polymerization, and cationic polymerization. This is because ethene does not have any substituent groups which influence the stability of the propagation head of the polymer. Each of these methods results in a different type of polyethylene.
Classification of polyethylenes
Polyethylene is classified into several different categories based mostly on its density and branching. The mechanical properties of PE depend significantly on variables such as the extent and type of branching, the crystal structure, and the molecular weight.
UHMWPE (ultra high molecular weight PE)
HDPE (high density PE)
HDXLPE (high density cross-linked PE)
PEX (cross-linked PE)
MDPE (medium density PE)
LDPE (low density PE)
LLDPE (linear low density PE)
VLDPE (very low density PE)
UHMWPE is polyethylene with a molecular weight numbering in the millions, usually between 3.1 and 5.67 million. The high molecular weight results in less efficient packing of the chains into the crystal structure as evidenced by densities less than high density polyethylene (e.g. 0.935 - 0.930). The high molecular weight results in a very tough material. UHMWPE can be made through any catalyst technology, although Ziegler catalysts are most common. UHMWPE is used in high modulus fibers and in bulletproof vests.
HDPE is defined by a density of greater or equal to 0.941 g/cc. HDPE has a low degree of branching and thus stronger intermolecular forces and tensile strength. HDPE can be produced by chromium/silica catalysts, Ziegler-Natta catalysts or metallocene catalysts. The lack of branching is ensured by an appropriate choice of catalyst (e.g. Chromium catalysts or Ziegler-Natta catalysts and reaction conditions.
PEX is a medium- to high-density polyethylene containing cross-link bonds introduced into the polymer structure, changing the thermoplast into an elastomer. The high-temperature properties of the polymer are improved, its flow is reduced and its chemical resistance is enhanced.
MDPE is defined by a density range of 0.926 - 0.940 g/cc. MDPE can be produced by chromium/silica catalysts, Ziegler-Natta catalysts or metallocene catalysts.
LLDPE is defined by a density range of 0.915 - 0.925 g/cc. is a substantially linear polymer, with significant numbers of short branches, commonly made by copolymerization of ethylene with short-chain alpha-olefins (e.g. 1-butene, 1-hexene, and 1-octene.
LDPE is defined by a density range of 0.910 - 0.940 g/cc. LDPE has a high degree of short and long chain branching, which means that the chains do not pack into the crystal structure as well. It has therefore less strong intermolecular forces as the instantaneous-dipole induced-dipole attraction is less. This results in a lower tensile strength and increased ductility. LDPE is created by free radical polymerization. The high degree of branches with long chains gives molten LDPE unique and desirable flow properties.
VLDPE is defined by a density range of 0.880 - 0.915 g/cc. is a substantially linear polymer, with high levels of short chain branches, commonly made by copolymerization of ethylene with short-chain alpha-olefins (e.g. 1-butene, 1-hexene, and 1-octene. VLDPE is most commonly produced using metallocene catalysts due to the greater co-monomer incorporation exhibited by these catalysts.
The most common household use of HDPE is in containers for milk, liquid laundry detergent, etc.; the most common household use of LDPE is in plastic bags. LLDPE is used in flexible tubing and in bags either neat or blended with LDPE.
HDPE is also widely used in the fireworks community. In tubes of varying length (depending on the size of the ordinance), HDPE is used as a replacement for the supplied cardboard mortar tubes for two primary reasons. One, it is much safer than the supplied cardboard tubes because if a shell were to malfunction and explode inside (flower pot) an HDPE tube, the tube will not shatter. The second reason is that they are reusable allowing designers to create multiple shot mortar racks. All pyrotechnicians discourage the use of PVC tubing in mortar tubes because it will shatter, sending shards of plastic at possible spectators, and will not show up in x-rays.
Recently, much research activity has focused on the nature and distribution of Long Chain Branches in polyethylene. These branches are present in all polyethylenes to some degree, and are very common in LDPE. In HDPE however, a relatively small number of these branches (perhaps 1 in 100 or 1000 branches per backbone carbon) can significantly affect the rheological properties of the polymer.
History
Polyethylene was first synthesized by the German chemist Hans von Pechmann, who prepared it by accident in 1898 while heating diazomethane. When his colleagues Eugen Bamberger and Friedrich Tschirner characterized the white, waxy substance he had created, they recognized that it contained long -CH2- chains and termed it polymethylene.
The first industrially practical polyethylene synthesis was discovered (again by accident) by Eric Fawcett and Reginald Gibson at ICI Chemicals in 1933. Upon applying extremely high pressure (several hundred atmospheres) to a mixture of ethylene and benzaldehyde, they again produced a white waxy material. Since the reaction had been initiated by trace oxygen contamination in their apparatus, the experiment was at first difficult to reproduce. It was not until 1935 that another ICI chemist, Michael Perrin, developed this accident into a reproducible high-pressure synthesis for polyethylene that became the basis for industrial LDPE production beginning in 1939.
Subsequent landmarks in polyethylene synthesis have centered around the development of several types of catalyst that promote ethylene polymerization at more mild temperatures and pressures. The first of these was a chromium trioxide based catalyst discovered in 1951 by Robert Banks and John Hogan at Phillips Petroleum. In 1953, the German chemist Karl Ziegler developed a catalytic system based on titanium halides and organoaluminum compounds that worked at even milder conditions than the Phillips catalyst. The Phillips catalyst is less expensive and easier to work with, however, and both methods are used in industrial practice.
By the end of the 1950s both the Phillips and Ziegler type catalysts were being used for HDPE production. Phillips' initially had difficulties producing a HDPE product of uniform quality, and filled warehouses with off-specification plastic. However, financial ruin was unexpectedly averted in 1957, when the hula hoop, a toy consisting of a circular polyethylene tube, became a fad among teenagers throughout the United States.
A third type of catalytic system, one based on metallocenes, was discovered in 1976 in Germany by Walter Kaminsky and Hansjrg Sinn. The Ziegler and metallocene catalyst families have since proven to be very flexible at copolymerizing ethylene with other olefins and have become the basis for the wide range of polyethylene resins available today, including VLDPE, and LLDPE. Such resins, in the form of fibers like Dyneema, have (as of 2005) begun to replace aramids in many high-strength applications.
Until recently, the metallocenes were the most active single-site catalysts for ethylene polymerisation known - new catalysts are typically compared to zirconocene dichloride. Much effort is currently being exerted on developing new single-site (so-called post-metallocene) catalysts, that may allow greater tuning of the polymer structure than is possible with metallocenes. Recently, work by Fujita at the Mitsui corporation (amongst others) has demonstrated that certain salicylaldimine complexes of Group 4 metals show substantially higher activity than the metallocenes.
Physical properties
Depending on the crystallinity and molecular weight, a melting point and glass transition may or may not be observable. The temperature at which these occur varies strongly with the type of PE.
| The above article is licensed under the GNU Free Documentation License. From Wikipedia, the free encyclopedia (https://en.wikipedia.org/wiki/polyethylene). Modified by Apparel Search 12/22/05. |